All pathology results have been resulted.
All screening windows have closed.
And eligible patients from 2024 have been randomized.
Even though it’s already the beginning of March, we’re excited to dive into some of the learnings from Summit Clinical Research in 2024.
As many of you may know 2024 was incredibly challenging for the Summit family with the passing of our co-founder, Dr. Stephen A. Harrison. Despite our sadness we are excited about the ways that we are investing in Dr. Harrison‘s legacy. Dr. Harrison never slowed down in life and would surely want to honor this momentum as caretakers of the companies he founded. We are eternally grateful to our families, our friends, site staff, and employees for the support you’ve shown us. We look forward to sharing more with you this year.
In contrast, 2024 was also a monumental year with the launch of the first US FDA-approved MASH drug, Rezdiffra® (resmetirom) from Madrigal. Summit Clinical Research was honored to play a large role in the development of this new drug. This launch is an exciting moment for the patients who may benefit from this treatment. It’s also an important pivot point for drug developers as they seek to understand the impact, if any, on their development plans. We expect to see further development of Rezdiffra in new patient populations for label expansion, and to better understand the experience of this drug in the real world offering important insights into liver and metabolic disease.
Figure 1
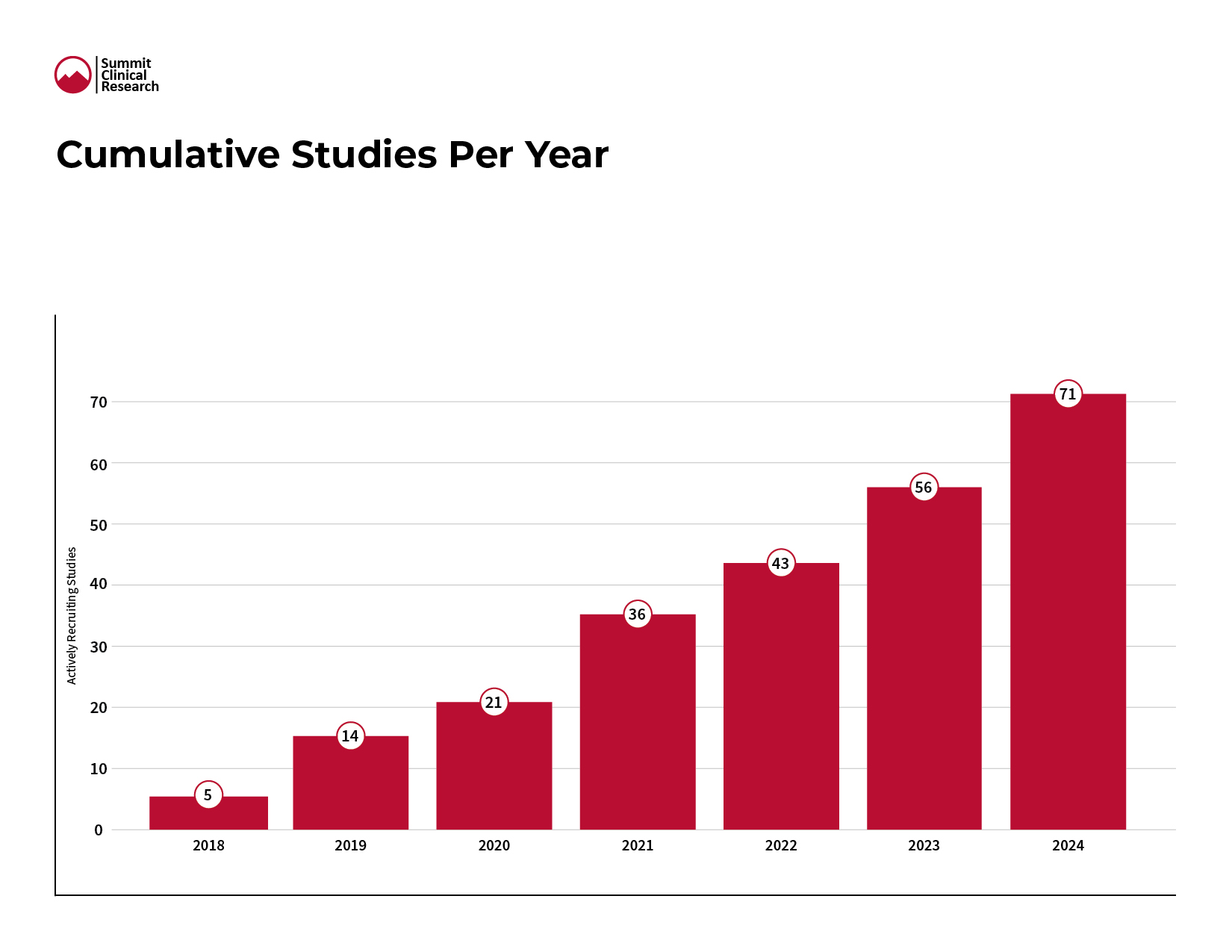
Summit Clinical Research initiated 15 new trials in 2024 bringing our total to 71 clinical trials since company inception in 2018 (Figure 1). These trials certainly bring new treatment options to patients in their respective disease states. They also create potential competition among drug developers and this topic is one of our most common points of engagement with sponsors in the metabolic and liver disease space. Given the breadth and depth of Summit’s involvement with these trials we have key insights to aid in site selection, site engagement, patient enrollment, patient retention, and overall trial success.
Though Summit experienced a 14% percent growth in new trials from last year, and not represented in the image, this count of new trials is lower than industry expectations. Anecdotally, these shifts appear to be delayed starts rather than cancellations, and 2025 is already off to a busy start with new trials. We’re tracking a few key trends in this “stutter step” we saw for new trial starts in 2024:
- Harrison’s untimely passing: Dr. Harrison was an accelerant for the entire therapeutic area. He acted as KOL and/or medical advisor for myriad sponsors; he helped sponsors network with Summit sites; he encouraged Summit sites to expand and grow (such as Pinnacle Clinical Research growing to 11 locations); he connected sponsors with Summit Clinical Research to execute the best trials; and he laid the foundation for innovation in the MASH space for Summit to perpetuate.
- Commercial launch of Rezdiffra: the US FDA approval of Rezdiffra (resmetirom) is monumental, and along with the adoption of the new drug is the requisite shifting of other development programs as sponsors figure out what, if anything, will happen to their programs. This launch also brings added visibility to the MASH disease area which has a halo effect to other MASH development programs with the hope that prescribers and patients are more aware of the disease.
- Adoption of GLP-1: Recent estimates are 1 in 8 patients in the US are using a GLP-1 treatment. These treatments have shown weight loss efficacy for some patients and also are studied across the metabolic spectrum including Type 2 Diabetes, MASH, and now Alzheimer’s Disease. These drugs offer compelling combination opportunities in these diseases. However, drug developers must navigate how to evaluate the efficacy of their drugs against GLP-1, as well as determine how to consider GLP-1 exposure at the patient level.
As we face this challenging landscape together, what challenges and opportunities do you see? Get in touch with Summit…

